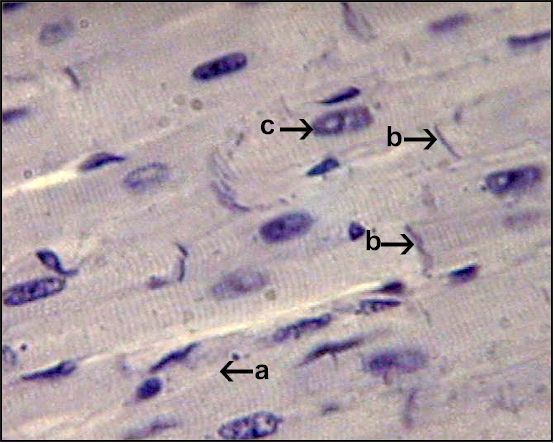
4, 5įigure 25-1 Schematic illustration of interactions of identified molecules comprising the fully coupled-pacemaker cell clock. These coupled oscillators that differ in nature thus represent heterogeneous redundancy within SANC that confers a physiological robust, yet extremely flexible, united pacemaker cell system. 3 During spontaneous AP firing by SANC, the M- and Ca 2+-clocks do not exist as separate entities: They mutually entrain each other via aforementioned coupling factors. Thus, similar to the M-clock, the SR is intrinsically also an almost perfectly periodic oscillator that can be envisioned as a Ca 2+-clock. 4 SANC sarcoplasmic reticulum (SR), however, in isolation from the M-clock can generate its own spontaneous local (submembrane) rhythmic Ca 2+ signals. 2 During rhythmic spontaneous AP firing by SANC, the M-clock is dynamically coupled to Ca 2+ cycling (see Figure 25-1, gray area) via multiple voltage-, Ca 2+-, cyclic adenosine monophosphate (cAMP)-, and phosphorylation-dependent mechanisms (coupling factors). 2 The subsystem of sarcolemmal molecules forming a voltage membrane oscillator was conceptualized as a membrane clock 3 (M-clock, for short) and has been the predominant feature in at least 12 SANC numerical models. Sarcolemmal ion currents (Figure 25-1 and Table 25-1) have been extensively studied in the past (see review 1), and the ensemble of these currents re-created in silico from experimental voltage-clamp data can generate spontaneous APs. The present perspective views SANC, per se, as a system comprising several levels of complexity and integrated components. The interactive network of mechanisms intrinsic to SANC must also interpret and react to signals arising extrinsic to the cell (e.g., stretch, electrotonic impulses, neurotransmitter or hormonal stimulation of surface membrane receptors). SANC robustness (i.e., “fail safe” properties conserved during evolution of the animal kingdom) and flexibility (the ability to react to demands for faster or slower AP firing rate) result from the timely integration of signaling events at multiple levels within the SAN, including subcellular, cellular surface membrane, and tissue architecture. DD represents a slow spontaneous decrease in membrane polarization until it reaches the membrane excitation threshold, culminating in generation of an AP. The essence of cardiac pacemaker function is diastolic depolarization (DD) that is absent in ventricular myocytes. 
Thus, SANC normally initiate the cardiac impulse by generating spontaneous APs that are conducted to the ventricle and entrain the duty cycle of ventricular myocytes. SAN cells (SANC) are the dominant cardiac pacemaker cells, because they exhibit shorter periods between spontaneous APs than do atrioventricular nodal cells or His-Purkinje cells. Pacemaker cells within the heart having “clocks” with the briefest rhythmic periods “capture” or trigger other excitable cells. 
The heart’s pacemaker cells normally generate spontaneous rhythmic changes in their membrane potential, thereby producing almost perfectly periodic spontaneous action potential (APs i.e., normal automaticity). The Essence of Pacemaker Function Is a Coupled Function of an Intracellular Ca 2+-Clock and Membrane Ion Channel ClockĪutonomic Receptor Modulation of the Coupled-Clock SystemĪn Additional Level of Complexity of Heart Pacemaker Function Arises Within the SAN Tissueĭifferent types of heart cells (i.e., pacemaker cells within the sinoatrial node and contractile myocytes within myocardium) determine how fast and strong, the heart beats. Realization of Importance of Ca 2+ Signaling for Cardiac Pacemaker FunctionĬrosstalk of Ryr and NCX to Transfer Intracellular Ca 2+ Signals to M-Clock
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
